  
Overall, though, Arc shader is a fantastic choice for gamers who want to enhance the visual appeal of their game without compromising performance. This mod’s drawbacks include the incompatibility with earlier versions of Minecraft and the potential impact on framerate if it is set too high. A depth-of-field effect can also be added to give the world a more realistic appearance. This realistic shaderpack provides new lighting effects, shadows, and reflections, among other things that may be customized to alter the game’s appearance. 
Arc Shaders are designed to enhance the game’s graphics without significantly slowing down performance. Therefore, whether you’re an experienced player or a newcomer, make sure to look at some of the greatest Minecraft shaders and see what they can do for you. Additionally, new shaders are always being produced, so there is always something fresh to test. They are therefore a crucial component of every player’s toolkit in Minecraft. Additionally, shaders can ease eye strain and make it simpler to see threats. The game becomes more immersive as a result, in addition to being more visually beautiful. Shaders give the game’s graphics a lot more realism by incorporating shadows and other effects. Minecraft shaders have significantly improved the fun factor of the game over time. Trust us, if you get this amazing modification tool, you will be a happy person because of the all features that It gives you. It is not that hard to download Optifine, you can find a great tutorial on our website on this topic. Thanks to this, you will adjust the settings to your needs and get best FPS as possible. It offers so much new options and new settings to the game. It will not only give you a shaderpack, you will also get a huge performance boost along with much more FPS. We believe that the Optifine is much better option for you. As you might know, if you want to get Minecraft Shaders, you need to have Optifine or the Iris Shaders. So, while keeping a high degree of performance, Minecraft shaders provide a fantastic approach to enhance the game’s visual appeal. However, they can also make their shaders operate more smoothly on a variety of hardware, allowing gamers to take advantage of the improved aesthetics without sacrificing performance. We’re wishing you luck! On the one hand, shader creators can concentrate on making their works appear as realistic as possible, with accurate lighting and shadows that give the virtual environment a sense of reality. If you don’t, you’ll overlook some important information and be unable to evaluate the author’s entire body of work. You only need to gather your ideas and take your time. The pack ensures a lot of pleasant emotions by attempting to provide realistic images with very little performance loss. Install Arc for Minecraft as soon as possible its new features will undoubtedly please you. Arc Shaders is a sizable shader pack that urges you to embark on a thrilling trip and take in the game’s fresh setting. The ability of Minecraft shaders to balance performance and aesthetics is one of its most amazing features. Whatever shader you have, you can install Arc Shader along with Optifine and Iris with confidence that it won’t adversely affect your game’s performance. Performance and realistic appearances are intended to be balanced by this shader. Arc Shaders 1.20.2 → 1.19 is a great new shaderpack for Minecraft. 
0 Comments
3/28/2024 0 Comments Minecraft house designs 
Unlike other houses, you will need to set up a special way of teleportation in Minecraft to reach it. Many pro players use such simple houses as their main bases during speedruns. The main purpose of a beginner house is to protect you from mobs spawning at night and nothing else. The best part about this house is that you can make it anywhere and in any style. It can be built within 5 minutes and will be as effective as a mansion while protecting you. You will find the blocks to make it at almost any spawn location. With that out of the way, let’s jump right into our first Minecraft house design on this list.įirst up, we have a simple wood and dirt-based house. We have marked each of the best Minecraft House ideas with a type to give you an idea of what to expect while constructing. Nevertheless, the end result is always something that makes you feel proud. These houses will require several blocks, including rare ones and a lot of dedicated time. Serious: We recommend you cheat a little and make the “serious” houses in creative game mode.Keep in mind that you might have to spend some time researching the perfect locations for some of these. They can be great if you plan to make multiple bases or just want some diversity in general. A few of the blocks used in these might take some time to find. Casual: Stepping towards houses that require a little bit of planning, these aren’t big on looks but definitely smart in strategy.You can make several of these throughout your world. They are great to make on the move and in sticky situations, where you need quick shelter. These house ideas do not require any unique blocks or much planning. Basic: Easy to build houses that can be made within an hour.So, to keep things simple, we are only classifying the houses based on the difficulty of their construction: And if we listen to the fandom, there are too many types of houses to keep a record of. Officially, there is are no specific types of houses in Minecraft, as they are constructed by the players only and can vary drastically based on their preferences. While creating a house, you can use one of our best Minecraft village seeds to make populating your villages an easier task. Such bases are only useful with the presence of useful villagers in them. Many players create a full-fledged base instead of a regular house, complete with villagers with assigned jobs. They are also a great way to collect free loots.Īs an added bonus, you can even use the house to populate the traditionally empty Minecraft Biomes such as savanna and badlands. And if you are an experienced player, you can even create some amazing automatic farms around your house. Acting as a safe house, you can use it to avoid mobs and store thousands of blocks with the right system. It is nowhere near as fiddly as the others on the list and you don't need anywhere near as many materials either - so a perfect starter castle.Other than making your worlds look better, Minecraft houses are also a great place to store items and better manage your inventory. This is a great one to try if you want to start building castles but you are not quite ready to make the leap to the larger, more sprawling, designs that are out there.   
The ground state electronic configuration of C is 1s22s22p2, with two unpaired electrons in p orbital and one pair of electrons in s orbital of valence shell. Now, we will use valence bond theory to understand the hybridization of ethane. The hybridization term is used in the valence bond theory (VBT) to explain the shape, formation, and directional properties of bonds in polyatomic molecules. 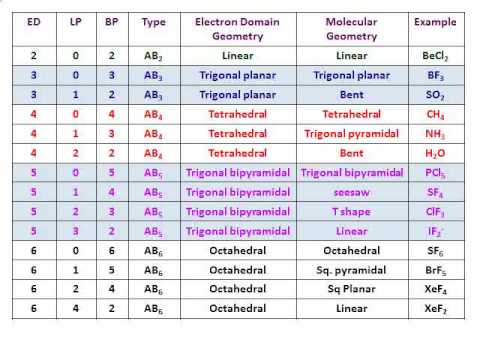
Hybridization is the mixing of two or more atomic orbitals of similar energy, which leads to the formation of hybrid orbitals. The bond length of the C-C and C-H bond is 153.52 pm and 109.40 pm, respectively.Īfter studying the lewis structure and molecular geometry, we move towards the hybridization of carbon atoms in the ethane molecule. The tetrahedral geometry of ethane leads to a bond angle (either H-C-H or H-C-H) of 109.5 °. Hence, the three-dimensional structure of ethane would be: The four bond pair of carbon corresponds to the tetrahedral geometry/shape of each carbon atom whereas one bond pair of every hydrogen atom corresponds to linear geometry/shape. We have observed already that carbon, a central atom forms 4 bonds, one with the nearest carbon atom and 3 with hydrogen atoms in the Lewis structure. So, the geometry/shape of ethane can be predicted from the following table: General formula The geometry and shape of ethane will be the same owing to the absence of lone pair of electrons. 
In ethane, carbon is a central atom and it has no lone pair of electrons. The molecular geometry of a compound is determined by valance shell electron pair repulsion (VSEPR) theory.Īccording to this theory, the shape and geometry of the molecule depend on the number of bonding electrons and lone pair of electrons. Now, let us move towards from 2D representation to 3D representation of the molecule i.e., molecular geometry of ethane. Therefore, there is no lone pair of electrons on any atom of ethane. If we observe, we can see that all 14 valance electron has been used for bond formation. Every hydrogen atom is surrounded by two electrons, leading to duplet formation. The total number of electrons around each carbon atom is 8 electrons and hence, it has completed its octet. We can count the total number of valance electrons in the Lewis structure of ethane, which is equal to 14. Therefore, it forms 4 bonds, one with neighboring carbon atoms and three with three hydrogen atoms by sharing electrons. In ethane, we have two carbon atoms and 6 hydrogen atoms and hence, the total number of valence electron are (2 X 4) + (1 X 6) = 14.Ī carbon atom has 4 valance electrons and it needs 4 more electrons to complete its octet. The valence electron for carbon (1s22s22p2) and hydrogen (1s1) is 4 and 1, respectively. Step 1: Determining the total number of valence electrons in the molecule. Let us draw a Lewis structure of ethane step by step. every atom tends to complete its octet ( 8 electrons) either by gaining or losing electrons except Hydrogen and Helium as they complete their duplet. Lewis structure is a 2D representation of the compound, which represents only the valance shell electrons of the atoms in the molecule. Let us discuss the basic concepts of ethane such as its Lewis structure, polarity, hybridization of carbon atom in ethane, and its Molecular orbital (MO) diagram to understand its chemical bonding in terms of molecular orbitals. 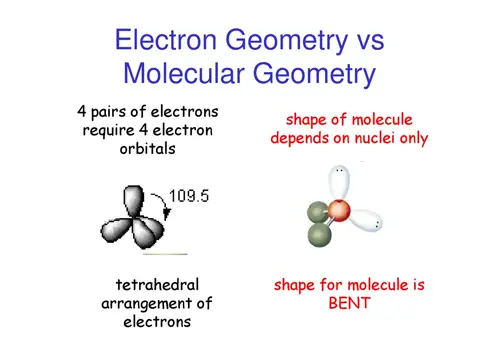
Here are some methods of preparation of ethane:ĬH3 − CH2Cl + H2 - Zn/H+ -–> CH3 − CH3 + HClĢCH3Cl + 2Na -Dry ether-> CH3 − CH3 + 2 NaClĢ CH3COONa + 2H2O -–Electrolysis-> CH3 − CH3 + 2NaOH + H2 + 2CO2 It can also be prepared from ethene, ethyl chloride, and sodium acetate in the laboratory. Ethane is obtained from natural gas and petroleum industrially. The flashpoint of ethane is -135 ☌ and hence, its vapors ignite easily by an ignition source. The melting and boiling point of ethane are -182.8 ☌ and -89 ☌, respectively. Ethane can also be written as CH3-CH3.Įthane is colorless and odorless gas at standard temperature and pressure. Saturated hydrocarbons are further classified into alkane (open chain of carbon atoms) and cycloalkane (closed chain of carbon atoms). Saturated hydrocarbons are those hydrocarbons, which contain carbon-hydrogen and carbon-carbon single bonds. Hydrocarbon is an organic compound, which contains only carbon and hydrogen. C2H6, known as ethane, is a saturated open-chain hydrocarbon or we can say that it comes under the alkane family.  3/28/2024 0 Comments Crime patrol satark online 
Maybe, that signal/feeling/instinct is just not enough to believe that it could result in a crime. Instincts/feelings/signals, that so often tell us that not everything is normal. Crime Patrol - Satark, will attempt to look at the signs, the signals that are always there before these mindless crimes are committed.  
Yet, when the heart takes over the mind, the outcome is a mindless tragedy. The cracks are there, too wide to be missed. Misunderstood, unresolved callings of the heart. Dont miss this thrilling and suspenseful episode of Crime Patrol Satark, only on Sony LIV. Watch the latest episode of Search, where a missing girls case leads to a shocking revelation. As the year passed by the woman started to face a lot of issues which made her life full of suffering. Crime Patrol Satark is a popular TV series that showcases real-life crime stories and investigations. It is difficult to comprehend the damage. A marriage that occurred through an online dating app. The husband is angry and the wife is upset. Crime Patrol Satark Full Episodes Episode 199 onwards. In a house, a husband and wife argue, fight. It is difficult to understand the intentions but the hints are there. That gaze, that quirky smile, that persistent stare which unnerves. Would knowing the Why? behind a crime, help in stopping a crime from happening? I don't like the way he looks at me, I don't like the way he/she is behaving, I think he/she is out of his/her mind, I think he/she has gone crazy. Every crime ignites a feeling that it should not have happened. Every crime we hear of, either warns us to be careful or scares us, that it could happen to us. Crimes that tell us lives could have been saved. sanjeev Tyagi and nissar khan Sanjiv Tyagi and nisar khan cases cr. Aapka Time shuru hota hai ab - Nissar Khan Crime Patrol. Crimes that tell us that we need to be careful, we need to be watchful. hot Crime patrol hot Episodes Crime Patrol 2020 latest episode N. Yet when the heart takes over the mind, the outcome is a mindless tragedy.Crime Patrol is back with it's 4th season, and attempts to bring stories of crime happening all around the country. The prelude and finale to the case would make the case a complete story. 
However the case presentation would be a story telling form that would have the interest of a fiction drama presentation. Misunderstood, unresolved callings of the heart. Crime Patrol attempts to bring stories of crime happening all around the country. It is difficult to comprehend the damage, but the cracks are there.Feelings. It is difficult to understand the intentions but the hints are there.In a house a husband and wife argue, fight. Every crime ignites a feeling, "It should not have happened".Would knowing the "Why" behind a crime, help in stopping a crime from happening?"I don't like the way he looks at me", "I don't like the way he/she is behaving", "I think he/she is out of his/her mind", "I think he/she has gone crazy". In Today’s episode, we will get to know that a young couple named Kajal and Abhishek are stabbed by few unidentified persons in the middle of the night. Crime Patrol Satark is a show that brings you the stories of real-life crimes and how they are solved by the police. Watch and Enjoy full episodes of Crime Patrol Satark (Bangla) on Sony LIV. Crimes that tell us lives could have been saved.Every crime we hear of, either warns us to be careful or scares us, it could happen to us. Watch Episode 668 Full Episode Online from Crime Patrol Satark (Bangla) only on Sony LIV. Crimes that tell us, we need to be careful, we need to be watchful. Samar succeed in stopping the unstoppable corruption which takes place in medical field?Ĭrime Patrol coming back in its 4th season attempts to bring stories of crime happening all around the country. Samar and forces Cops to reopen the case. Awasthi dies due to severe stroke and court shuts the case but this does not go too well with Dr. Samar to take his share and stay out of this matter but Dr. Awasthi who is a root cause of all evil bribes Dr. The police officials recovers a womans dead body. over billing thus he pressurizes Chief Medical officer V.N. In Todays episode, we present to you a brutal murder case which is executed in the heart of Mumbai. When the police investigation begins, the blame falls on Dimples ex-boyfriend. Ep 412 - Crime Patrol Satark - In this UP special episode of Crime Patrol Satark, Host Anoop Soni explores the story of 2 Families who go through the same pa. Samar working in Haripur Hospital gets a huge shock when he observes medicine reports where he comes to know that the medicines which are supplied in the hospital are charged 4 times higher than it's orignal price i.e. U/A 16+ Crime Hindi 2003 49minutes Dimple, an ambitious and confident girl, gets kidnapped by three goons, soon after her wedding.  3/28/2024 0 Comments Download shareit without app store 
SHAREit is further available to users as an entertainment platform where people can discover and stream a multitude of things. As if these features were not enough, the application even includes a feature of inbuilt music and videos.This app can come in handy when you want to let a person know of your exact location. You may even need to share your location for other reasons as well. 
You can share your location with your friends and family and let them know your whereabouts. Regardless of which, if you are amongst those who’re concerned about their personal safety, then definitely go for SHAREit as it provides a solution for that as well.
 
This group consists of trigonal planar molecules and bent molecules. All linear molecules share the property that they are completely straight i.e the bond angle from one bond to another is Electron Groups Notice that there will be several different linear geometries. Linear geometries occur when there are only 2 bonds and 0 lone pairs. This group consists only of the linear geometry. Now let's go over the geometries in terms of the number of electron pairs: From all outward appearances, they will look identical however. For example, both molecules with 2 bonds and 0 lone pairs and 2 bonds and 3 lone pairs will be linear. They will have different numbers of bonds and lone pairs, but the end result will be the same. One brief thing to take note of is that there are multiple linear and bent geometries. You'll see that the chart follows a very straightforward pattern of logic. The molecular geometry chart may seem like a lot of information at first, but over time it will become intuitive. The molecular geometry for a water molecule is bent, which is why the water molecule isn't a straight H-O-H molecule. If we look up at the table, we can find on the left side a column for molecules with 4 electrons groups, and within that group a molecule with 2 lone pairs. The central `O` atom has 4 groups of electrons around it: 2 bonds and 2 unpaired electrons, also called lone pairs. Let's say you had the formula of water, `H_2O`, and wanted to know its molecular geometry. In other words, by drawing out the Lewis structure of a molecule, one can determine the molecule's 3d orientation. We're going to discuss each one individually, but note that you can determine the molecular geometry of a molecule solely by the number of bonds and lone pairs around the central atom. The following chart provides the different molecular geometries and the conditions in which they arise. This will become important when discussing bond angles. If you substitute a single bond with a lone pair, the lone pair will repel the other electron groups away from it more than the single bond would. 
One key point is that lone pairs take up more room than single bonds do. The lone pair is usually not shown in 3d models and thus you have to visualize it yourself. This is why `NH_3` has a tri-pod shape: the lone pair on top of `N` pushes the other electrons (in the bond) away from it. The basic idea behind VSEPR is that electron pairs will repel each other. The only difference is the number of electrons on the central atom: `N` has a lone pair whereas `B` does not. Take a look at the Lewis structures of `NH_3` and `BF_3`. Molecular geometry is determined by the arrangement of valence electrons. The reason behind this is that the two structures have a different number and arrangement of valence electrons. This is the case of `BF_3` and `NH_3`: both have the general formula `AB_3`, but `NH_3` on the left is a tri-pod shape whereas `BF_3` on the right is a 3-legged starfish shape. In other words, two molecules with the general formulas `AB_3` may look completely different in real life: one may be a pyramid whereas the other may be completely flat. Valence Shell Electron Pair Repulsion (VSEPR) is a theory that states that the 3d orientation, also known as the molecular geometry, of a molecule is not dependent on its chemical formula but on the repulsion of valence electrons.  3/24/2024 0 Comments Oneida dispatch obituaries 
and Todd Newland of East Syracuse six brothers and five sisters-in-law, Russell and Lee Chase of Earlville, Chester and Mary Chase of Taberg, Marvin and Linda Chase of Norwich, David and Amber Chase of Mt. Chase and Stephanie Welke of Chittenango two daughters and a son-in-law, Cheryl A. Surviving are his wife three sons and a daughter-in-law, Bryan E. He had been a 4-H leader for many years, and loved hunting, fishing, reading and playing cards. in New York Mills for 9 years, and also did carpentry work in the area for many years. Formerly he had worked for Hubbell Highway Sign Co. 
He had worked in the building maintenance department at the Canastota Central Schools for almost 20 years, retiring in 1996. Ernie spent his early years in the New Berlin area, attending the Morris and Guilford Schools, living in Canastota since 1959. 7, 1933, in Pittsfield, the son of Claude C. “Ernie” Chase, 67, of 2929 State Route 31, died Friday, March 23, 2001, at his home following a lengthy illness. and Kathy Vredenburg of Higginsville one daughter and son-in-law, Linda and Harold “Butch” Shingler of Oriskany Falls one step-daughter and step-son-in-law, Nancy and Niles “Butch” Hawver of Clockville one brother, John Vredenburg of Vienna one sister and brother-in-law, Betty Jane and Duane Holmes of Blossvale 11 grandchildren 12 great-grandchildren one great-great-grandchild and several nieces and nephews. Surviving are his wife, Eleanor two sons and one daughter-in-law, Harry R. He was a member of Laborers International Union of North America, Local No. Vredenburg was employed at Hamilton College and previously was self-employed in the construction industry. Prior to his retirement in 1985, with 17 years of service, Mr. 
19, 1946, in Hickory Knolls, Oneida Castle. A resident of the Oriskany Falls area for the past 37 years, Mr. He was born on May 30, 1922, in Rome, the son of Harry and Mabel Owens Vredenburg. Vredenburg, 78, of 5907 Tanner Road, died Thursday evening, June 15, 2000, in the Oneida Healthcare Center, where he had been a patient for the past week. and Donna and Mark La Londe of Oneida one additional brother-in-law, Leonard Nichols of Rome and several nieces and nephews. her father-in-law and mother-in-law, Clarence “Pete” and Clara Nichols of Wampsville two sisters-in-law and one brother-in-law, Marie Nichols of Laurel, Md. Morgan, all of Bouckville, and Alfred and Ellie Morgan, of North Bay five sisters and four brothers-in-law, Dorothy and Ray Gunther of Hamilton, Phyllis and Charles Faulkner of Nicholasville, Ky., Barbara and Tom Springer of Madison, Diane and Bill Gunthorpe of Morrisville, and Janet Morgan Whiteman of New Martinsville, W. Nichols, a student at Cazenovia College four brothers and three sisters-in-law, Eugene and Judy Morgan of Los Lunas, N. Nichols Jr., at home one daughter, Kristen I. Patrick’s Church, where she was active with several church groups and participated on various softball leagues, including the league for the area’s churches. Nichols was employed with Colgate University, in the athletics department. Nichols was a graduate of Hamilton High School and attended Mohawk Valley Community College. 
A resident of the Oneida area for the past twenty-one years, Mrs. 26, 1946, in Bouckville, the daughter of Emerson P. Nichols, 54, of 397 Genesee St., died Saturday, June 17, 2000, in the Oneida Healthcare Center, where she had been a medical patient for the past four days, following a lengthy illness.   In addition to her husband, she was preceded in death by her sisters, Henrietta Schroeder and Mary Margaret Gayman and her sons-in-law, Brian L. and Jeni Utter, Brad and Erin Utter and Scott and Lindy Utter fourteen great-grandchildren and two great-great-grandchildren and her sister, Gayle Meier of Austin, Texas. Those left to honor her memory include her daughters, Judith Jones of Davenport and Jill Utter of Durant, Iowa her grandchildren, Todd and Val Jones, Amy and Mike Archer, Matt Jones, Brian C. In earlier years, she and Jack enjoyed frequent trips to Las Vegas and New Orleans. Friends and family have shared their relationship to show their support. We welcome you to provide your thoughts and memories on our Tribute Wall. An obituary is not available at this time for Stephen Lyons. Gerri enjoyed playing cards with her family, looked forward to dinner out at the Family Square Restaurant, adored her grandkids and loved dark chocolate. Public viewing will be Thursday 11/09 from 11-5 at Jones Unity Funeral Home. Send Sympathy Gifts Plant a Tree for Stephen. Funeral services will be held on Saturday Novem11:00 at. Timothy Stallworth will be held on Friday, Novemfrom 11-5 at Jones Unity Funeral Home. She was a 35-year volunteer “Pink Lady” at Genesis Medical Center, East Campus and a former member of St. Obituary for Timothy Stallworth Visitation for Mr. Burial will be at Bright Morning Star Baptist Church Cemetery in Frisco City, AL. Funeral services will be Friday 09/01 12 at Bell Ree Enterprise in Frisco City, AL. 
Gerri was a teller at the Brenton Bank, Northwest Bank and the American Bank of Rock Island where she retired many years ago. Public viewing will be Thursday 08/31 from 11-5 at Jones Unity Funeral Home. He preceded her in death on August 18, 2011. On September 3, 1949, she was united in marriage to Jack L. She was born in Davenport on August 13, 1929, the daughter of Henry and Sylvia (Krejci) Bormann. Memorials may be made to Unity Point Hospice. Burial will be in Davenport Memorial Park. 
AJ and Isabelle Frye in Monroe County Alabama. on Tuesday, Jat the McGinnis-Chambers Funeral Home in Bettendorf. Barbara Jean Frye Dale, 82 was born on Jto the late Andrew J. Monroeville in Monroeville, AL. He departed this life much too soon on January 17,2024 at the age of 40 years old.įather: Donald Eugene Hurst, Sr., Dallas, TXĬhildren: Ke’Rra Wherry, Latroya Hurst, Jermaine Neal, Ziyana Hurst, Khileigh Hurst, Mikhialo Smith, Grandson – Kaymdon Brown.Īunts: Ruby Mack, Elizabeth Stephens, Rolanda White, Demetress Henderson, Cynthia HendersonĪ host of other loving cousins, family and friends.Funeral services to celebrate the life of Geraldine B. Stephen Lyonss passing on Thursday, Decemhas been publicly announced by Jones Unity Funeral Home Inc. He was hard worker and a well-respected employee of Daniels Feed Store, Dekalb, TX. He especially was a loving father who gave his last, sacrificing himself for his children. Banks Public viewing will be 06/23 from 11-5 at Jones Unity Funeral Home. Gene was a kind soul with a loving spirit to all he met. Offer condolences/tributes, send flowers or create an online memorial for free. Search Unity obituaries and condolences, hosted by. He graduated from Atlanta High School in 2001. Obituaries from Jones Unity Funeral Home Inc. He was preceded in death by his mother and a nephew Jordan Mims. Grand children, Richard Williams, Shaundrika Collins, Rolandon Morine, Karmesha Morine, Trenell Moore, Demontric Martin, Zaria Dunn, Bryson Martin, Jaima Colbert, James Colbert, Jaheim Callahan, Demarcus Callahan, Ranneshia Callahan, along with 19 Great Grandchildren and a host of other relatives and friends.ĭonald Eugene (Gene) Hurst, Jr. Timothy Stallworths passing at the age of 43 on Friday, Novemhas been publicly announced by Jones Unity Funeral Home Inc. 
She leaves to cherish her memories forever, her two sons, Billy Callahan of Texarkana, Arkansas and Roderick Dunn of Little Rock, Arkansas Special Son, Charles Moore, of Dallas, Texas her four daughters, Minister Sonya Collins, of Texarkana, Arkansas Donna Williams, British Dunn-Brown (Briant), and Bernadette Dunn-Martin (Lamont) all of Texarkana Texas. Cora Sue Johnson Barkley was born Novemin Finchburg, AL to the late Andy (Shug) Johnson and Sally Shoots Johnson. Dunn was proceeded in death by her parents, Hoyle Callahan and Josephine Callahan, and siblings: Albert Griffin, Eugene Holmes, Earnestine Thomas, Sue Ella (Monica) Sauls, and Hoyle Callahan, Jr. Dunn retired from Defense Logistics Agency at Red River Army Depot in June 2023 after 43 years of dedicated service. OUR DEEPEST CONDOLENCES TO THE KIDD FAMILY. VIEWING WILL BE HELD ON FRIDAY, APRIL 2 FROM 11-5 PM. 
Bertha Mae Callahan-Dunn, 76, of Texarkana, Arkansas, accepted her eternal wings on Sunday, January 28, 2024. JANNIE KIDD WILL BE HELD ON SATURDAY, APRIL 3RD AT 2:30 PM AT PINE FLAT BAPTIST CHURCH IN HYBART, AL.   Simply make a column for all the s orbitals with each n shell on a separate row. This chart is straightforward to construct. The arrow leads through each subshell in the appropriate filling order for electron configurations. Figure 3 illustrates the traditional way to remember the filling order for atomic orbitals. 
Electrons enter higher-energy subshells only after lower-energy subshells have been filled to capacity. Each added electron occupies the subshell of lowest energy available (in the order shown in Figure 1), subject to the limitations imposed by the allowed quantum numbers according to the Pauli exclusion principle. This procedure is called the Aufbau principle, from the German word Aufbau (“to build up”). Beginning with hydrogen, and continuing across the periods of the periodic table, we add one proton at a time to the nucleus and one electron to the proper subshell until we have described the electron configurations of all the elements. To determine the electron configuration for any particular atom, we can “build” the structures in the order of atomic numbers. The diagram of an electron configuration specifies the subshell ( n and l value, with letter symbol) and superscript number of electrons. The notation 3 d 8 (read “three–d–eight”) indicates eight electrons in the d subshell (i.e., l = 2) of the principal shell for which n = 3. A superscript number that designates the number of electrons in that particular subshell.įor example, the notation 2 p 4 (read “two–p–four”) indicates four electrons in a p subshell ( l = 1) with a principal quantum number ( n) of 2.The letter that designates the orbital type (the subshell, l), and.The number of the principal quantum shell, n,.We describe an electron configuration with a symbol that contains three pieces of information (Figure 2): The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We will discuss methods for remembering the observed order. For small orbitals (1 s through 3 p), the increase in energy due to n is more significant than the increase due to l however, for larger orbitals the two trends are comparable and cannot be simply predicted. Electrons in orbitals that experience more shielding are less stabilized and thus higher in energy. This phenomenon is called shielding and will be discussed in more detail at a later time. Electrons that are closer to the nucleus slightly repel electrons that are farther out, offsetting the more dominant electron–nucleus attractions slightly (recall that all electrons have −1 charges, but nuclei have + Z charges). In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s p > d > f. The energy of atomic orbitals increases as the principal quantum number, n, increases. Ch3.1 Orbital Energies and Atomic Structure The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. We can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Chapter 3: Electron Configurations and the Periodic Table  
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |




 RSS Feed
RSS Feed
